The human nail is an exceptionally strong barrier due to its keratinized structure and extensive disulfide bond cross-linking. As a result, drug delivery through the nail is highly challenging. Many ungual products require nail filing prior to application, and treatments often require longer contact times compared to other topical dosage forms.
Penetration enhancement strategies may include:
MedPharm has experience developing a range of ungual dosage forms, including:
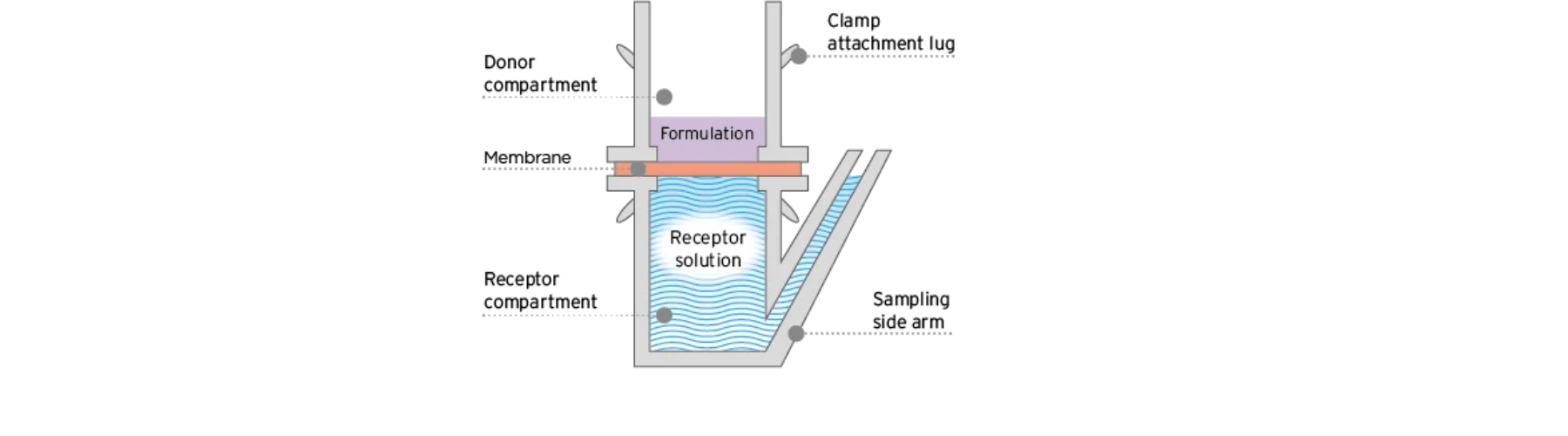
MedPharm’s Nail IVPT model uses the proprietary ChubTur® static diffusion cell, specifically designed for permeation and penetration studies through hard tissues such as nails.
Human nail clippings are secured using a custom gasket that prevents lateral migration of formulation, ensuring accurate measurement of drug movement through nail tissue.
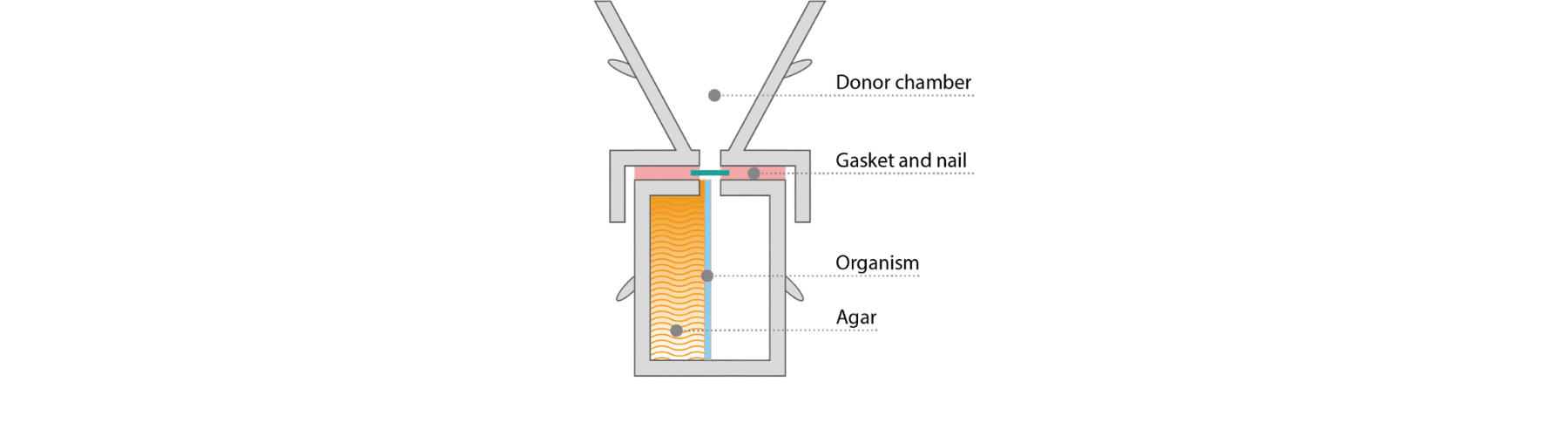
The Nail Zone of Inhibition assay uses MedPharm’s proprietary TurChub® diffusion cell and is based on agar zone-of-inhibition principles.
Human nails, including sections, whole clippings, cadaver nails, and diseased nails, are mounted above agar inoculated with bacteria, yeast, or fungi. Formulations are applied topically, and efficacy and permeation are assessed by the resulting zone of inhibition.
This approach provides semi-quantitative data without the need for analytical methods, enabling multiple drugs to be screened simultaneously while reducing development time and cost.
This biological assay combines MedPharm’s ChubTur® diffusion cell with active fungal infection to closely mimic clinical onychomycosis.
Fungi are cultured on the underside of healthy human nails to recreate clinically relevant infection. Formulations are applied to the nail surface, and viable organisms are measured using a modified ATP assay to determine percent viability or kill rate.
This model closely reflects the clinical condition and has been used as a biowaiver in place of clinical trials.


